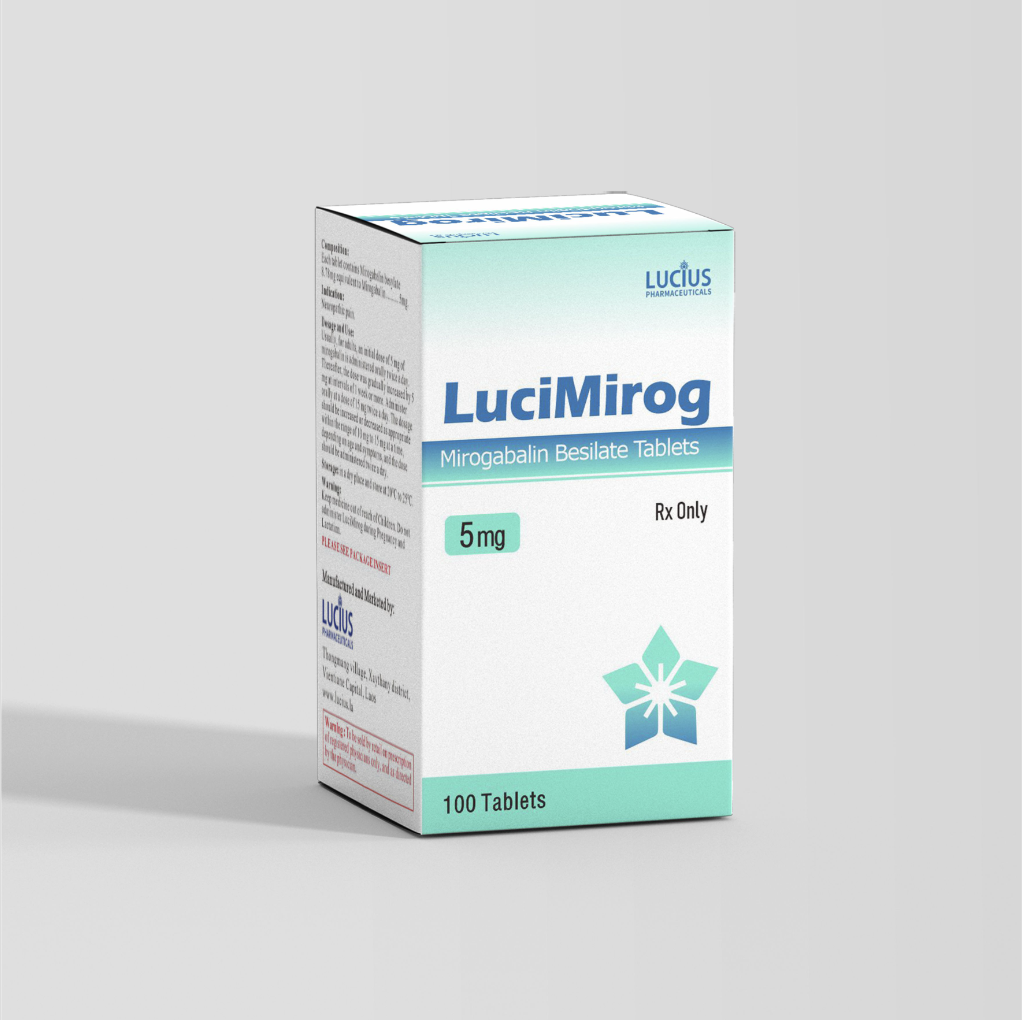
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use LuciMirog safely and effectively. See full prescribing information for LuciMirog.
INDICATIONS AND USAGE
Neuropathic pain.
DOSAGE AND ADMINISTRATION
Usually, for adults, an initial dose of 5 mg of mirogabalin is administered orally twice a day. Thereafter, the dose was gradually increased by 5 mg at intervals of 1 week or more. Administer orally at a dose of 15 mg twice a day. The dosage should be increased or decreased as appropriate within the range of 10 mg to 15 mg at a time, depending on age and symptoms, and the dose should be administered twice a day.
DOSAGE FORMS AND STRENGTHS
Tablets: 5 mg×100 tablets
CONTRAINDICATIONS
Patients with a history of hypersensitivity to the components of this drug.
WARNINGS AND PRECAUTIONS
• Dizziness, somnolence, loss of consciousness, etc. may occur, so patients receiving this drug should be careful not to engage in dangerous activities such as driving a car.
• This may cause weight gain, so be careful of obesity, and if signs of obesity appear, take appropriate measures such as diet and exercise therapy. In particular, as weight gain may be observed with increased dosage or long-term administration, the patient's weight should be measured periodically.
• Since the treatment of neuropathic pain with this drug is a symptomatic treatment rather than a causative treatment, the disease that causes the pain should be diagnosed and treated at the same time, and this drug should not be administered carelessly.
• Sudden discontinuation of administration of this drug may cause withdrawal symptoms such as insomnia, nausea, diarrhea, and loss of appetite; therefore, when discontinuing administration, proceed carefully by gradually reducing the dose.
• Eye disorders such as amblyopia, visual abnormalities, blurred vision, and double vision may occur, so be careful to ask questions regarding eye disorders during your examination.
ADVERSE REACTIONS
Serious side effects
Dizziness (frequency unknown), somnolence (frequency unknown), loss of consciousness (less than 0.1%).
There is a risk of falling and causing fractures, etc. If any abnormalities are observed, take appropriate measures such as discontinuing administration or reducing the dose.
Hepatic dysfunction (incidence unknown)
Hepatic dysfunction such as increased AST and ALT may occur. If any abnormalities are observed, including initial symptoms such as general malaise and loss of appetite, administration should be discontinued and appropriate measures should be taken.
USE IN SPECIFIC POPULATIONS
• Patients with renal dysfunction
Adjust the dosage and dosing interval based on the creatinine clearance value. The plasma concentration of this drug may increase, making side effects more likely.
• Pregnant woman
It should be administered to pregnant women or women who may become pregnant only if the therapeutic benefits are judged to outweigh the risks. Placental penetration has been reported in animal experiments (rats).
• Breastfeeding woman
Consider continuing or discontinuing breastfeeding, taking into account the therapeutic benefit and the benefit of breastfeeding. It has been reported that it migrates into milk in animal experiments (rats).
• Pediatric use
No clinical trials have been conducted on children.
• Senior citizen
Dosage and interval based on creatinine clearance value Administer carefully by adjusting the interval. Kidney function is often decreased. Dizziness, somnolence, Loss of consciousness may cause a fall and fracture.
Storage
Store at 20℃ to 25℃ (68℉ to 77℉), excursions permitted between 15℃ and 30℃ (59℉ and 86℉) [see USP Controlled Room Temperature]. Protect from moisture.